Efficacy and safety established in the ZUMA-1 pivotal trial and additional safety studies (Cohorts 4 and 6)1,2
ZUMA-1 study design1-4
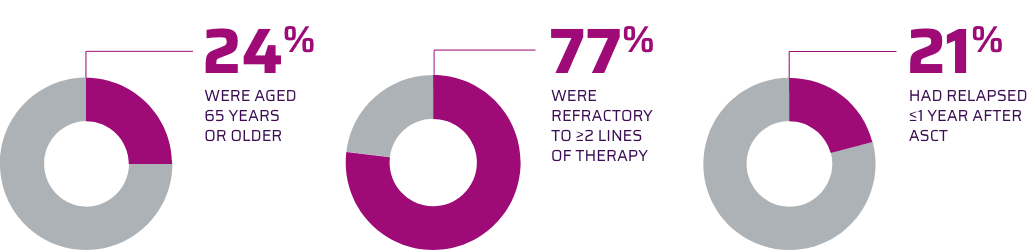
- YESCARTA was studied in a phase 2, open-label, single-arm, multicenter trial of 101 adults with relapsed/refractory (R/R) aggressive B-cell non-Hodgkin lymphoma, comprising the pivotal trial (Cohorts 1 and 2) upon which YESCARTA was FDA approved
- Patients had refractory disease to the most recent therapy or relapsed within 1 year after autologous stem cell transplantation (ASCT)
- Patients received lymphodepleting chemotherapy: 500 mg/m2 intravenous (IV) cyclophosphamide and 30 mg/m2 IV fludarabine (5, 4, and 3 days before infusion)
- YESCARTA was administered as a single infusion to a target dose of 2 x 106 viable CAR T cells/kg body weight (maximum of 2 x 108 viable CAR T cells)
- Bridging chemotherapy between leukapheresis and lymphodepleting chemotherapy was not permitted in Cohorts 1 and 2
- In the commercial setting, the use of bridging therapy after leukapheresis and before lymphodepleting chemotherapy is at the discretion of the treating physician
Cohorts 4 and 61,2,5,6
- The first safety study (Cohort 4) was a subsequent, open-label, safety-management cohort of 46 patients with large B-cell lymphoma (LBCL), 41 of whom were treated with YESCARTA to assess the early use of corticosteroids and/or tocilizumab for Grade 1 cytokine release syndrome (CRS) or neurologic toxicity
- Optional bridging chemotherapy was permitted in Cohort 4
- The second safety-management study (Cohort 6) was a second subsequent open-label, safety-management cohort of 39 patients with LBCL, investigating the effects of prophylactic corticosteroid and levetiracetam use + earlier corticosteroid and/or tocilizumab intervention on the incidence of Grade 1 CRS and neurologic toxicities in YESCARTA-treated patients with R/R LBCL
- Optional bridging chemotherapy was permitted in Cohort 6
YESCARTA delivers durable, complete remissions1
*Data from the 27.1-month median follow-up of ZUMA-1 are not included in the Prescribing Information (PI) for YESCARTA and should be carefully interpreted.8,9
†The ITT population included all 111 patients leukapheresed in the phase 2 segment of ZUMA-1; efficacy assessments were based on data from the mITT population (N=101) comprising all patients enrolled and treated with the target dose of YESCARTA. ITT data are not included in the PI for YESCARTA.1,10
Subgroup response rates7§
Data from the investigator analysis of ZUMA-1 are based on assessments performed by individual investigators at their respective trial sites and should be interpreted with caution.7 Investigator-assessed data are not included in the PI for YESCARTA. For FDA approval, efficacy was established on the basis of CR rate and DOR, as determined by an IRC.1
§Subgroup data not included in the full PI. The efficacy in these subgroups was not a study objective and the study was not powered to assess efficacy in these subgroups.
||Post hoc subgroup analysis.7
Many patients in Cohorts 1 and 2 achieved durable, complete responses3
The tick marks represent censored patients. Patients who did not progress, have not died, or received new anti-cancer therapy (including stem cell transplant) prior to documented progression were censored at their last evaluable disease assessment.14
Data from the 27.1-month median follow-up of ZUMA-1 are not included in the PI for YESCARTA and should be carefully interpreted.9
- At the time of FDA approval, the median follow-up was 11.6 months.4 Efficacy was established on the basis of CR rate and DOR, as determined by an IRC.1
5 and alive with YESCARTA: 43% of patients alive at 5 years with a median OS of 25.8 months17
The only CAR T to present overall survival (OS) data with at least 5 years of follow-up17,18
- OS was a secondary endpoint of the ZUMA-1 phase 2, single-arm, open-label study1,3
- OS data are descriptive and should be carefully interpreted in light of the single-arm design.3,12 OS data are not included in the Prescribing Information for YESCARTA
- Not all data continued to be captured at the 5-year follow-up. OS, investigator-assessed response, and adverse event reporting were captured19
- The KM estimate of the 5-year OS rate was 43%17

The tick marks represent censored patients. Patients who have not died by the data cutoff date were censored at the last date known to be alive.14
KM median OS: 25.8 months (95% CI, 12.8-NE).17
Updated safety-management studies (Cohort 4 and Cohort 6)
CAR T=chimeric antigen receptor T cell; CI=confidence interval; DLBCL=diffuse large B-cell lymphoma; DOR=duration of response; FDA=Food and Drug Administration; HGBCL=high-grade B-cell lymphoma; IRC=independent review committee; ITT=intent-to-treat; KM=Kaplan-Meier; mITT=modified intent-to-treat; mOS=median overall survival; NE=not estimable; NR=not reached; PMBCL=primary mediastinal large B-cell lymphoma; TFL=transformed follicular lymphoma.
IMPORTANT SAFETY INFORMATION
WARNING: CYTOKINE RELEASE SYNDROME, NEUROLOGIC TOXICITIES, and SECONDARY HEMATOLOGICAL MALIGNANCIES
- Cytokine Release Syndrome (CRS), including fatal or life-threatening reactions, occurred in patients receiving YESCARTA. Do not administer YESCARTA to patients with active infection or inflammatory disorders. Treat severe or life-threatening CRS with tocilizumab or tocilizumab and corticosteroids.
- Neurologic toxicities, including fatal or life-threatening reactions, occurred in patients receiving YESCARTA, including concurrently with CRS or after CRS resolution. Monitor for neurologic toxicities after treatment with YESCARTA. Provide supportive care and/or corticosteroids as needed.
- T cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T cell immunotherapies, including YESCARTA.
- YESCARTA is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the YESCARTA and TECARTUS REMS.
CYTOKINE RELEASE SYNDROME (CRS)
CRS, including fatal or life-threatening reactions, occurred following treatment with YESCARTA. CRS occurred in 90% (379/422) of patients with non-Hodgkin lymphoma (NHL), including ≥ Grade 3 CRS in 9%. CRS occurred in 93% (256/276) of patients with large B-cell lymphoma (LBCL), including ≥ Grade 3 in 9%. Among patients with LBCL who died after receiving YESCARTA, 4 had ongoing CRS events at the time of death. For patients with LBCL in ZUMA-1, the median time to onset of CRS was 2 days following infusion (range: 1-12 days) and the median duration was 7 days (range: 2-58 days). For patients with LBCL in ZUMA-7, the median time to onset of CRS was 3 days following infusion (range: 1-10 days) and the median duration was 7 days (range: 2-43 days).
CRS occurred in 84% (123/146) of patients with indolent non-Hodgkin lymphoma (iNHL) in ZUMA-5, including ≥ Grade 3 CRS in 8%. Among patients with iNHL who died after receiving YESCARTA, 1 patient had an ongoing CRS event at the time of death. The median time to onset of CRS was 4 days (range: 1-20 days) and median duration was 6 days (range: 1-27 days) for patients with iNHL.
Key manifestations of CRS (≥ 10%) in all patients combined included fever (85%), hypotension (40%), tachycardia (32%), chills (22%), hypoxia (20%), headache (15%), and fatigue (12%). Serious events that may be associated with CRS include, cardiac arrhythmias (including atrial fibrillation and ventricular tachycardia), renal insufficiency, cardiac failure, respiratory failure, cardiac arrest, capillary leak syndrome, multi-organ failure, and hemophagocytic lymphohistiocytosis/macrophage activation syndrome (HLH/MAS).
The impact of tocilizumab and/or corticosteroids on the incidence and severity of CRS was assessed in 2 subsequent cohorts of LBCL patients in ZUMA-1. Among patients who received tocilizumab and/or corticosteroids for ongoing Grade 1 events, CRS occurred in 93% (38/41), including 2% (1/41) with Grade 3 CRS; no patients experienced a Grade 4 or 5 event. The median time to onset of CRS was 2 days (range: 1-8 days) and the median duration of CRS was 7 days (range: 2-16 days). Prophylactic treatment with corticosteroids was administered to a cohort of 39 patients for 3 days beginning on the day of infusion of YESCARTA. Thirty-one of the 39 patients (79%) developed CRS and were managed with tocilizumab and/or therapeutic doses of corticosteroids with no patients developing ≥ Grade 3 CRS. The median time to onset of CRS was 5 days (range: 1-15 days) and the median duration of CRS was 4 days (range: 1-10 days). Although there is no known mechanistic explanation, consider the risk and benefits of prophylactic corticosteroids in the context of pre-existing comorbidities for the individual patient and the potential for the risk of Grade 4 and prolonged neurologic toxicities.
Ensure that 2 doses of tocilizumab are available prior to YESCARTA infusion. Monitor patients for signs and symptoms of CRS following infusion at least daily for 7 days at the certified healthcare facility, and for 4 weeks thereafter. Counsel patients to seek immediate medical attention should signs or symptoms of CRS occur at any time. At the first sign of CRS, institute treatment with supportive care, tocilizumab, or tocilizumab and corticosteroids as indicated.
NEUROLOGIC TOXICITIES
Neurologic toxicities including immune effector cell-associated neurotoxicity syndrome (ICANS) that were fatal or life-threatening occurred following treatment with YESCARTA. Neurologic toxicities occurred in 78% (330/422) of patients with NHL receiving YESCARTA, including ≥ Grade 3 in 25%.
Neurologic toxicities occurred in 87% (94/108) of patients with LBCL in ZUMA-1, including ≥ Grade 3 in 31% and in 74% (124/168) of patients in ZUMA-7 including ≥ Grade 3 in 25%. The median time to onset was 4 days (range: 1-43 days) and the median duration was 17 days for patients with LBCL in ZUMA-1. The median time to onset for neurologic toxicity was 5 days (range: 1-133 days) and median duration was 15 days in patients with LBCL in ZUMA-7. Neurologic toxicities occurred in 77% (112/146) of patients with iNHL, including ≥ Grade 3 in 21%. The median time to onset was 6 days (range: 1-79 days) and the median duration was 16 days. Ninety-eight percent of all neurologic toxicities in patients with LBCL and 99% of all neurologic toxicities in patients with iNHL occurred within the first 8 weeks of YESCARTA infusion. Neurologic toxicities occurred within the first 7 days of infusion in 87% of affected patients with LBCL and 74% of affected patients with iNHL.
The most common neurologic toxicities (≥ 10%) in all patients combined included encephalopathy (50%), headache (43%), tremor (29%), dizziness (21%), aphasia (17%), delirium (15%), and insomnia (10%). Prolonged encephalopathy lasting up to 173 days was noted. Serious events, including aphasia, leukoencephalopathy, dysarthria, lethargy, and seizures occurred. Fatal and serious cases of cerebral edema and encephalopathy, including late-onset encephalopathy, have occurred.
The impact of tocilizumab and/or corticosteroids on the incidence and severity of neurologic toxicities was assessed in 2 subsequent cohorts of LBCL patients in ZUMA-1. Among patients who received corticosteroids at the onset of Grade 1 toxicities, neurologic toxicities occurred in 78% (32/41) and 20% (8/41) had Grade 3 neurologic toxicities; no patients experienced a Grade 4 or 5 event. The median time to onset of neurologic toxicities was 6 days (range: 1-93 days) with a median duration of 8 days (range: 1-144 days). Prophylactic treatment with corticosteroids was administered to a cohort of 39 patients for 3 days beginning on the day of infusion of YESCARTA. Of those patients, 85% (33/39) developed neurologic toxicities; 8% (3/39) developed Grade 3, and 5% (2/39) developed Grade 4 neurologic toxicities. The median time to onset of neurologic toxicities was 6 days (range: 1-274 days) with a median duration of 12 days (range: 1-107 days). Prophylactic corticosteroids for management of CRS and neurologic toxicities may result in higher grade of neurologic toxicities or prolongation of neurologic toxicities, delay the onset, and decrease the duration of CRS.
Monitor patients for signs and symptoms of neurologic toxicities following infusion at least daily for 7 days at the certified healthcare facility, and for 4 weeks thereafter, and treat promptly.
REMS
Because of the risk of CRS and neurologic toxicities, YESCARTA is available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) called the YESCARTA and TECARTUS REMS. The required components of the YESCARTA and TECARTUS REMS are:
- Healthcare facilities that dispense and administer YESCARTA must be enrolled and comply with the REMS requirements. Certified healthcare facilities must have on-site, immediate access to tocilizumab, and ensure that a minimum of 2 doses of tocilizumab are available for each patient for infusion within 2 hours after YESCARTA infusion, if needed for treatment of CRS.
Further information is available at www.YescartaTecartusREMS.com or 1-844-454-KITE (5483).
HYPERSENSITIVITY REACTIONS
Allergic reactions may occur with the infusion of YESCARTA. Serious hypersensitivity reactions, including anaphylaxis, may be due to dimethyl sulfoxide (DMSO) or residual gentamicin in YESCARTA.
SERIOUS INFECTIONS
Severe or life-threatening infections occurred after YESCARTA infusion. Infections (all grades) occurred in 45% of patients with NHL. Grade 3 or higher infections occurred in 17% of patients, including ≥ Grade 3 infections with an unspecified pathogen in 12%, bacterial infections in 5%, viral infections in 3%, and fungal infections in 1%. YESCARTA should not be administered to patients with clinically significant active systemic infections. Monitor patients for signs and symptoms of infection before and after infusion and treat appropriately. Administer prophylactic antimicrobials according to local guidelines.
Febrile neutropenia was observed in 36% of patients with NHL and may be concurrent with CRS. In the event of febrile neutropenia, evaluate for infection and manage with broad-spectrum antibiotics, fluids, and other supportive care as medically indicated.
In immunosuppressed patients, including those who have received YESCARTA, life-threatening and fatal opportunistic infections including disseminated fungal infections (e.g., candida sepsis and aspergillus infections) and viral reactivation (e.g., human herpes virus-6 [HHV-6] encephalitis and JC virus progressive multifocal leukoencephalopathy [PML]) have been reported. The possibility of HHV-6 encephalitis and PML should be considered in immunosuppressed patients with neurologic events and appropriate diagnostic evaluations should be performed.
Hepatitis B virus (HBV) reactivation, in some cases resulting in fulminant hepatitis, hepatic failure, and death, has occurred in patients treated with drugs directed against B cells, including YESCARTA. Perform screening for HBV, HCV, and HIV and management in accordance with clinical guidelines before collection of cells for manufacturing.
PROLONGED CYTOPENIAS
Patients may exhibit cytopenias for several weeks following lymphodepleting chemotherapy and YESCARTA infusion. Grade 3 or higher cytopenias not resolved by Day 30 following YESCARTA infusion occurred in 39% of all patients with NHL and included neutropenia (33%), thrombocytopenia (13%), and anemia (8%). Monitor blood counts after infusion.
HYPOGAMMAGLOBULINEMIA
B-cell aplasia and hypogammaglobulinemia can occur in patients receiving YESCARTA. Hypogammaglobulinemia was reported as an adverse reaction in 14% of all patients with NHL. Monitor immunoglobulin levels after treatment and manage using infection precautions,
antibiotic prophylaxis, and immunoglobulin replacement.
The safety of immunization with live viral vaccines during or following YESCARTA treatment has not been studied. Vaccination with live virus vaccines is not recommended for at least 6 weeks prior to the start of lymphodepleting chemotherapy, during YESCARTA treatment, and until immune recovery following treatment.
SECONDARY MALIGNANCIES
Patients treated with YESCARTA may develop secondary malignancies. T cell malignancies have occurred following treatment of hematologic malignancies with BCMA- and CD19-directed genetically modified autologous T cell immunotherapies, including YESCARTA. Mature T cell malignancies, including CAR-positive tumors, may present as soon as weeks following infusion, and may include fatal outcomes.
Monitor life-long for secondary malignancies. In the event that a secondary malignancy occurs, contact Kite at 1-844-454-KITE (5483) to obtain instructions on patient samples to collect for testing.
EFFECTS ON ABILITY TO DRIVE AND USE MACHINES
Due to the potential for neurologic events, including altered mental status or seizures, patients are at risk for altered or decreased consciousness or coordination in the 8 weeks following YESCARTA infusion. Advise patients to
refrain from driving and engaging in hazardous occupations or activities, such as operating heavy or potentially dangerous machinery, during this initial period.
ADVERSE REACTIONS
The most common adverse reactions (incidence ≥ 20%) in patients with LBCL in ZUMA-1 included CRS, fever, hypotension, encephalopathy, tachycardia, fatigue, headache, decreased appetite, chills, diarrhea, febrile neutropenia, infections with pathogen unspecified, nausea, hypoxia, tremor, cough, vomiting, dizziness, constipation, and cardiac arrhythmias.
INDICATIONMORE
YESCARTA® is a CD19-directed genetically modified autologous T cell immunotherapy indicated for the treatment of:
- Adult patients with relapsed or refractory large B-cell lymphoma after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL) not otherwise specified, primary mediastinal large B-cell lymphoma, high grade B-cell lymphoma, and DLBCL arising from follicular lymphoma.
Limitations of Use: YESCARTA is not indicated for the treatment of patients with primary central nervous system lymphoma.
Please see full Prescribing Information, including BOXED WARNING and Medication Guide.
Authorized Treatment Centers are independent facilities certified to dispense Kite CAR T therapies. Choice of an Authorized Treatment Center is within the sole discretion of the physician and patient. Kite does not endorse any individual treatment sites.
References: 1. YESCARTA® (axicabtagene ciloleucel). Prescribing information. Kite Pharma, Inc; 2024. 2. Oluwole OO, Forcade E, Muñoz J, et al. Prophylactic corticosteroid use with axicabtagene ciloleucel in patients with relapsed/refractory large B-cell lymphoma: one-year follow-up of ZUMA-1 Cohort 6. Presented at: 63rd ASH Annual Meeting and Exposition; December 11-14, 2021; Atlanta, GA. Abstract 2832. 3. Locke FL, Ghobadi A, Jacobson CA, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1–2 trial. Lancet Oncol. 2019;20(1):31-42. 4. Locke FL, Ghobadi A, Jacobson CA, et al. Durability of response in ZUMA-1, the pivotal phase 2 study of axicabtagene ciloleucel (axi-cel) in patients (pts) with refractory large B-cell lymphoma. Presented at: 2018 ASCO Annual Meeting; June 1-5, 2018; Chicago, IL. Abstract 3003. 5. Topp MS, van Meerten T, Houot R, et al. Earlier steroid use with axicabtagene ciloleucel (axi-cel) in patients with relapsed/refractory large B cell lymphoma. Presented at: 61st ASH Annual Meeting and Exposition; December 7-10, 2019; Orlando, FL. Abstract 243. 6. Oluwole OO, Bouabdallah K, Muñoz J, et al. Prophylactic corticosteroid use in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br J Haematol. 2021;194(4):690-700. 7. Neelapu SS, Locke FL, Bartlett NL, et al. Axicabtagene ciloeucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med. 2017;377(26):2531-2544. 8. Data on file [1]. Kite Pharma, Inc; 2018. 9. Data on file [2]. Kite Pharma, Inc; 2018. 10. Data on file [3]. Kite Pharma, Inc; 2018. 11. Data on file. Kite Pharma, Inc; 2020. 12. Locke FL, Ghobadi A, Jacobson CA, et al. Long-term safety and activity of axicabtagene ciloleucel in refractory large B-cell lymphoma (ZUMA-1): a single-arm, multicentre, phase 1–2 trial. Lancet Oncol. 2019;20(1):31-42 (suppl). doi:10.1016/S1470-2045(18)30864-7 13. Data on file [4]. Kite Pharma, Inc; 2018. 14. Data on file [1]. Kite Pharma, Inc; 2019. 15. Data on file [5]. Kite Pharma, Inc; 2018. 16. Data on file [2]. Kite Pharma, Inc; 2019. 17. Jacobson CA, Locke F, Ghobadi A, et al. Long-term (4- and 5-year) overall survival in ZUMA-1, the pivotal study of axicabtagene ciloleucel in patients with refractory large B-cell lymphoma. Poster presented at: 63rd ASH Annual Meeting and Exposition; December 11-14, 2021; Atlanta, GA. Poster 1764. 18. Chong EA, Ruella M, Schuster SJ, et al. Five-year outcomes for refractory B-cell lymphomas with CAR T-cell therapy. N Engl J Med. 2021;384(7):673-674. 19. Data on file [3]. Kite Pharma, Inc; 2019. 20. Data on file. Kite Pharma, Inc; 2021. 21. Topp MS, van Meerten T, Houot R, et al. Earlier corticosteroid use for adverse event management in patients receiving axicabtagene ciloleucel for large B-cell lymphoma. Br J Haematol. 2021;195(3):388-398. 22. Data on file. Kite Pharma, Inc; 2022.